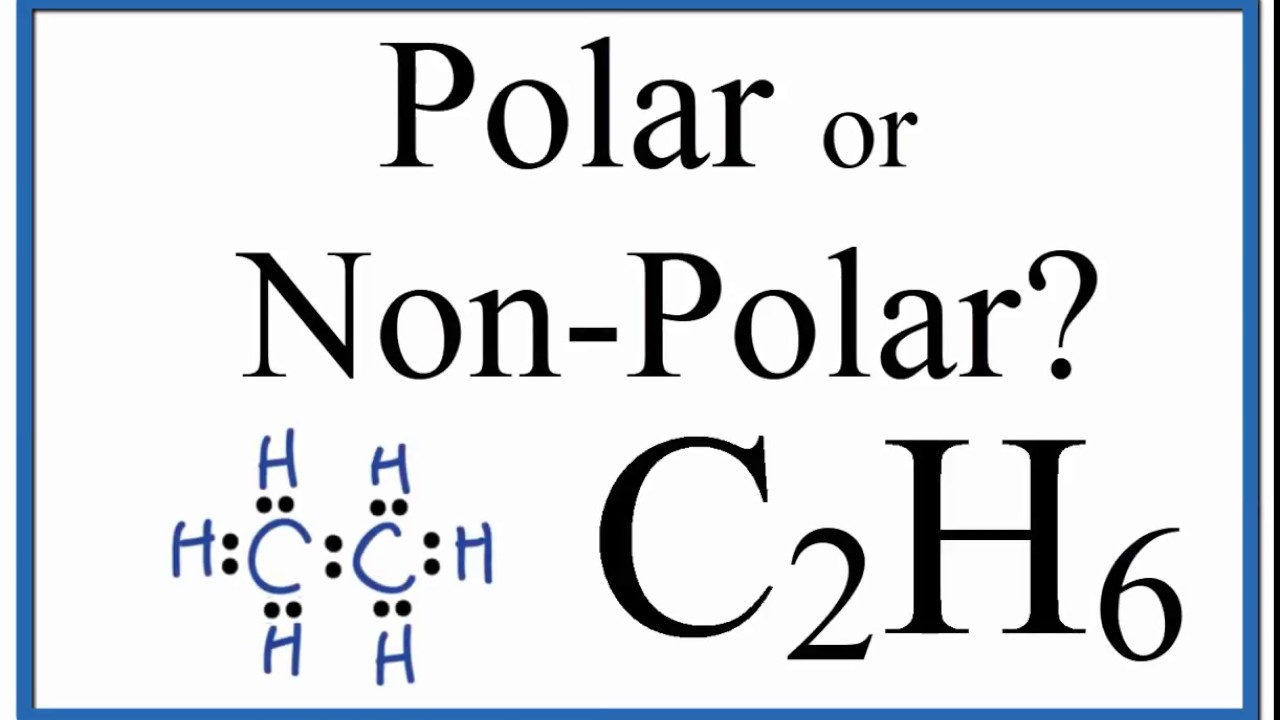BeH2 Lewis Structure (Beryllium Hydride) | BeH2 Lewis Structure (Beryllium Hydride) Beryllium Hydride or BeH2 has a simple arrangement of atoms. In this video, we help you find out the Lewis dot...

NELSON CHEMISTRY 12 SECTION 4.4 POLARITY What is the name of 007's Inuit cousin? Polar Bond. - ppt download

Review Polar Covalent: unequal sharing of electrons (electronegativity difference >0.4) Nonpolar Covalent: equal sharing of electrons (electronegativity. - ppt download

SOLVED: 2A) Which of the folloiwng molecules would be considered non-polar? BeH2, Br2S, PF3, TeCl2, HCL – show all work
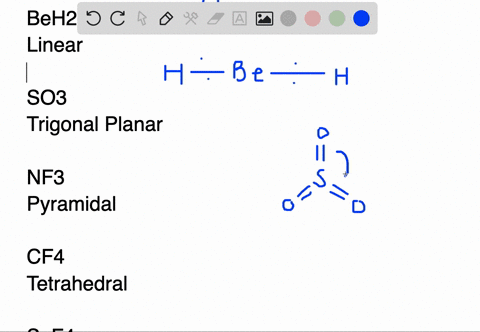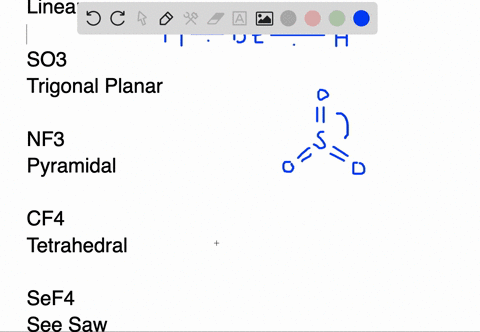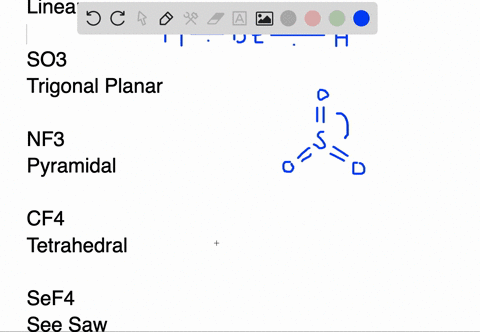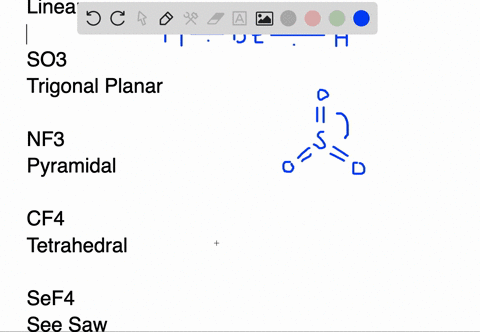
SOLVED:Draw Lewis structures and predict the molecular structures of the following. (See Exercises 89 and 90 . ) a. OCl2, KrF2, BeH2, SO2 c. CF4, SeF4, KrF4 b. SO3, NF3, IF3 d.

How many of the following compounds are non- polar? NH3 ,NF3 ,BeH2 ,CO2 ,H2O ,HI, H2 ,BF3 ,CH4 CHCl3 ,CH2Cl2 , CCl4