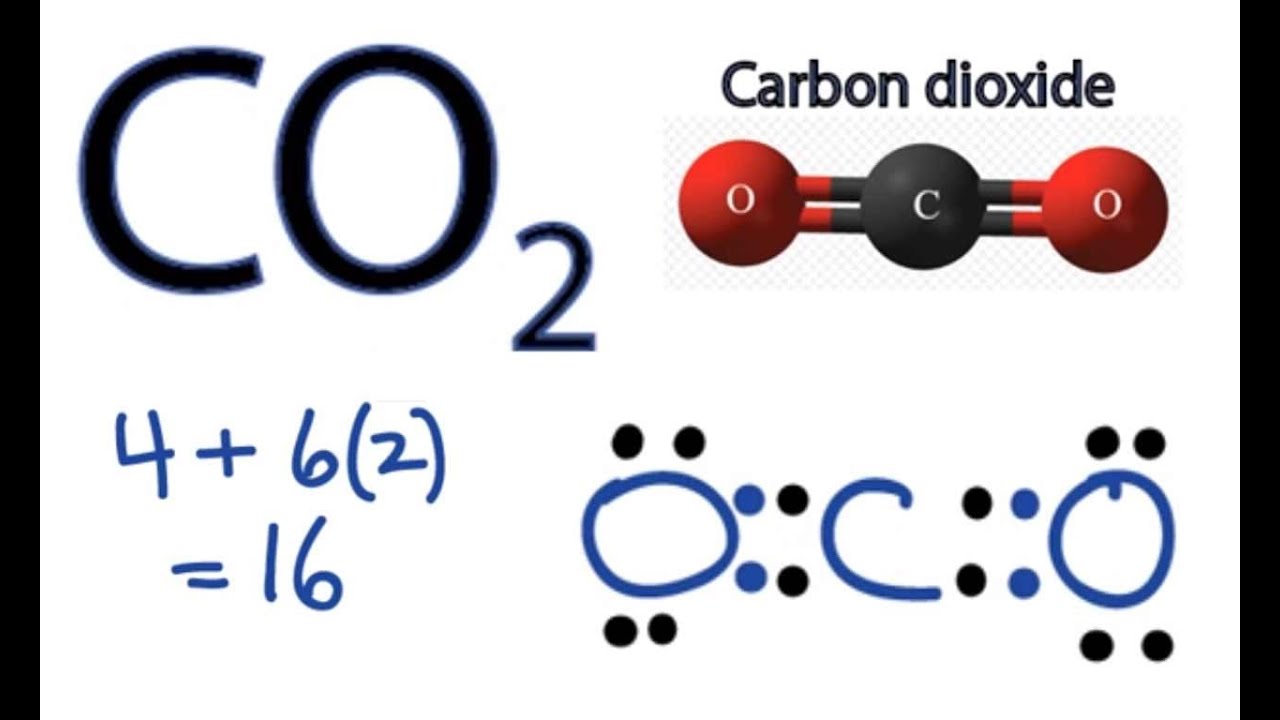Electrocatalytic Reduction of CO2 to Acetic Acid by a Molecular Manganese Corrole Complex - De - 2020 - Angewandte Chemie International Edition - Wiley Online Library

What is the simplest way for converting CO2 to O2? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

One-Pot Synthesis of Dimethyl Hexane-1,6-diyldicarbamate from CO2, Methanol, and Diamine over CeO2 Catalysts: A Route to an Isocyanate-Free Feedstock for Polyurethanes | ACS Sustainable Chemistry & Engineering
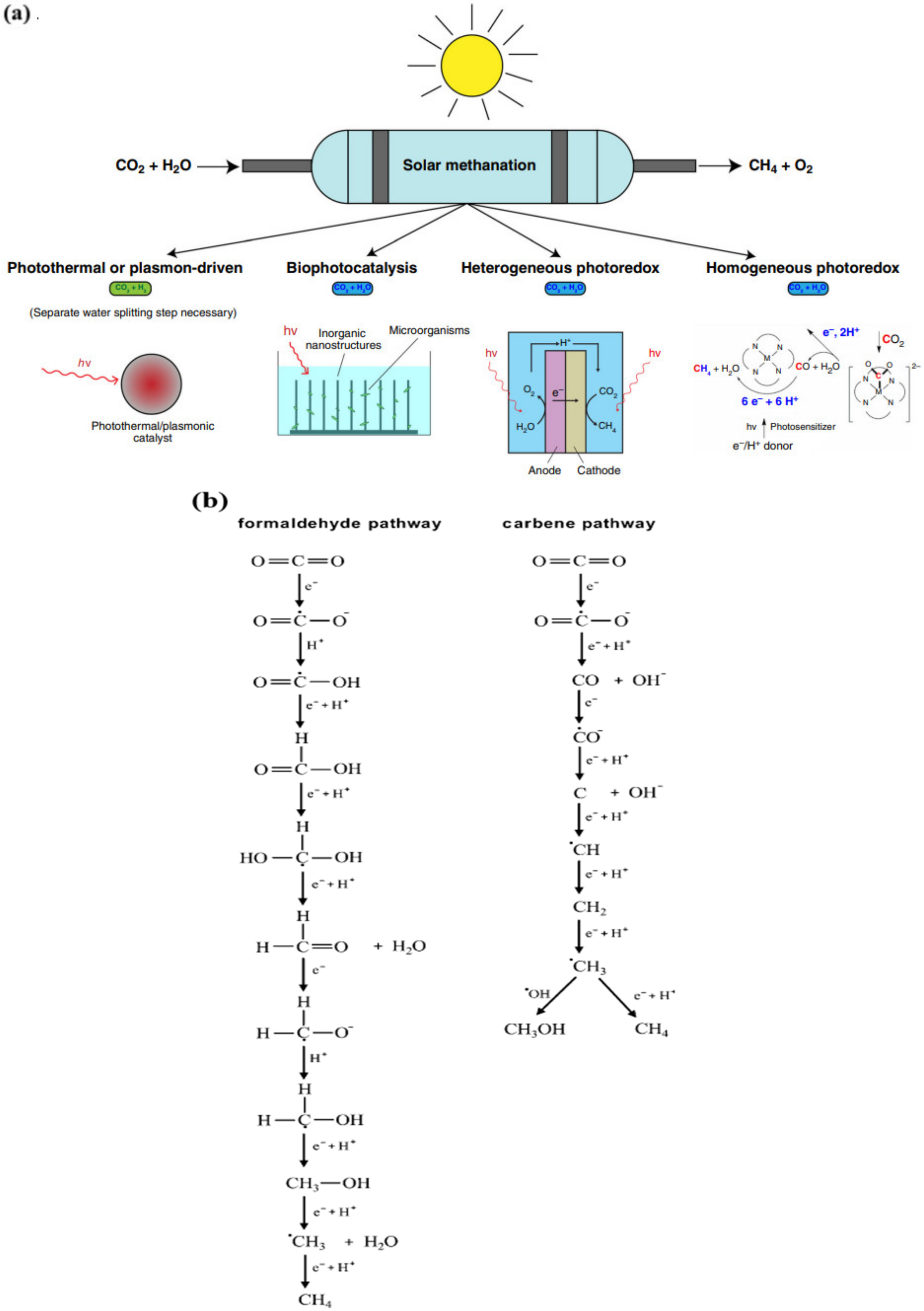
Catalysts | Free Full-Text | Direct Conversion of CO2 into Hydrocarbon Solar Fuels by a Synergistic Photothermal Catalysis
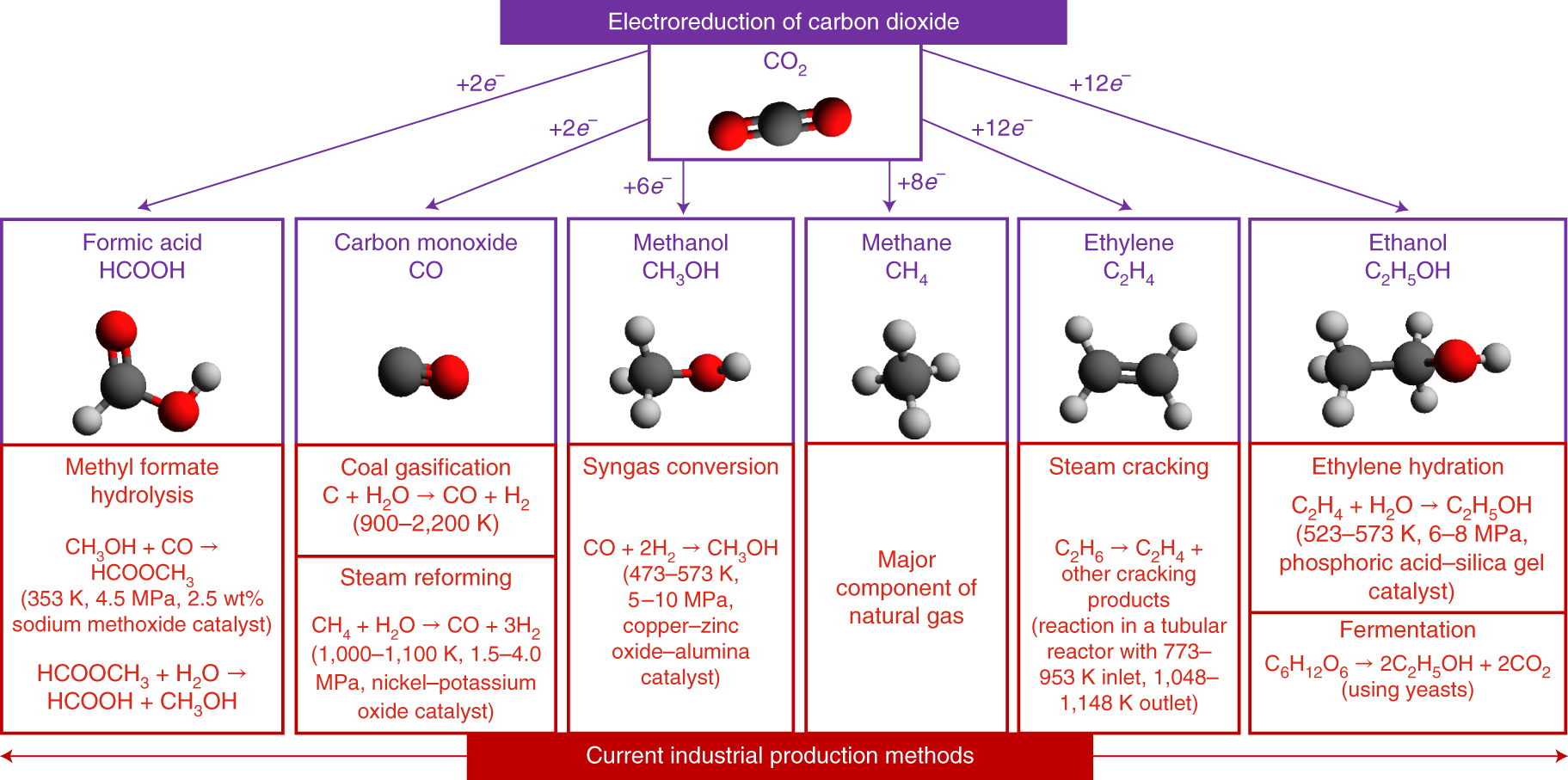
Co-electrolysis of CO2 and glycerol as a pathway to carbon chemicals with improved technoeconomics due to low electricity consumption | Nature Energy

Multimedia: Carbon Dioxide Can Make a Solution Acidic | Chapter 6, Lesson 10 | Middle School Chemistry

Amazon.com: Coldbreak Jockey Box Dispensing Kit, 1 Tap, Includes: CO2 Regulator, 4' Air Line, Sankey 'D' Beer Coupler (SS Probe), 6' Jockey Box Beverage Jumper (SS Tailpieces), NO Tank, SSDK1-D : Home

Consider this reaction: 6 CO2 + 6 H2O + light equation C6H12O6 + 6 O2 If there were 2.38 x 102 g of - Brainly.in
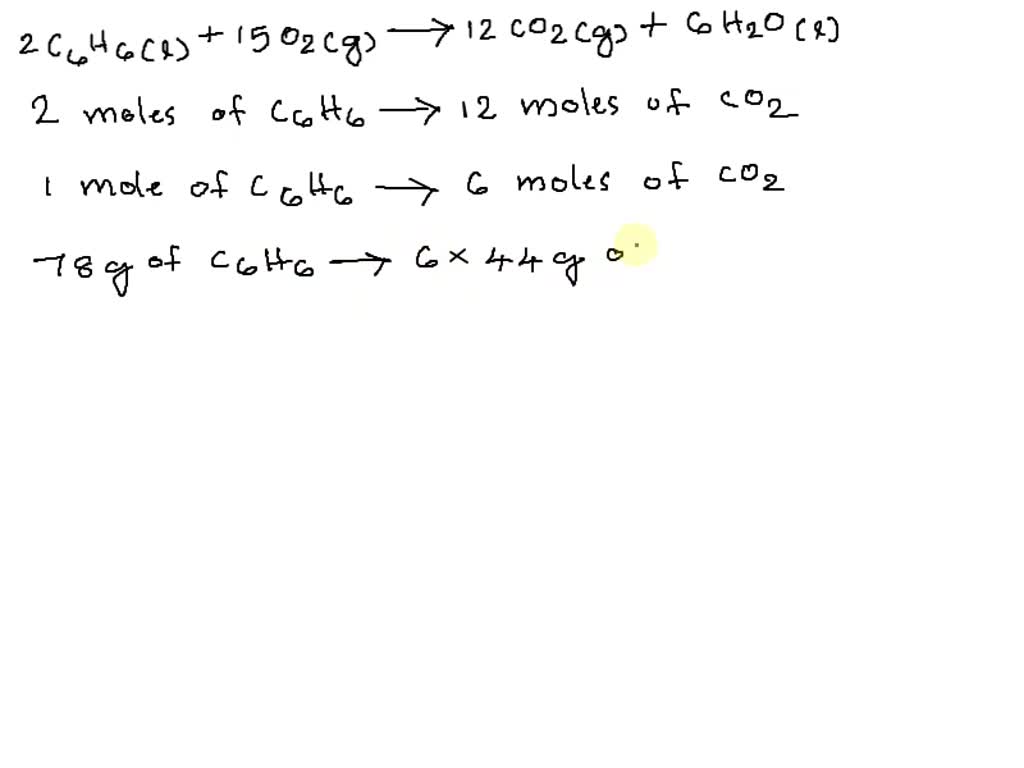







![BikeTiresDirect 16g Non-Threaded CO2 Cartridge (6-Pack) [42104-6] at BikeTiresDirect BikeTiresDirect 16g Non-Threaded CO2 Cartridge (6-Pack) [42104-6] at BikeTiresDirect](https://static.biketiresdirect.com/productimages/images450/vts5p1-1.jpg)