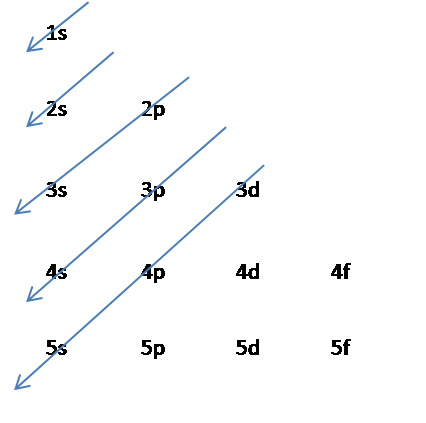
SOLVED:Write the condensed ground-state electron configuration for each of the following elements. The element's atomic number is given in parentheses. (a) \mathrm{Li}(3) (b) \mathrm{Ne}(10) (c) Be (4) (d) C(6) (e) \operatorname{Mg}(12)

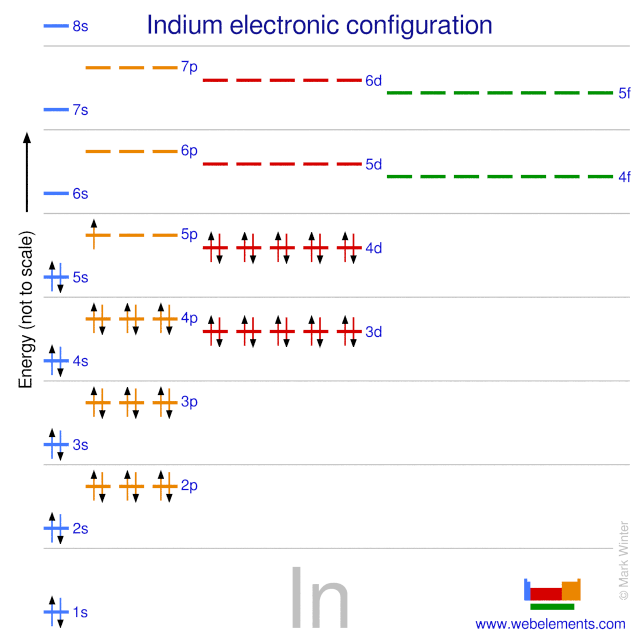
Electron Configuration - Detailed Explanation, Filling of orbital, Representation of Electronic Configuration of Atom with FAQs .

SOLVED:Write the condensed ground-state electron configuration for each of the following elements. The element's atomic number is given in parentheses. (a) \mathrm{Li}(3) (b) \mathrm{Ne}(10) (c) Be (4) (d) C(6) (e) \operatorname{Mg}(12)

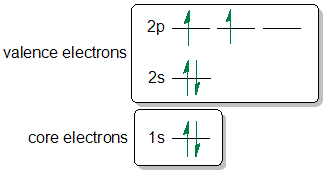
SOLVED:Draw a partial (valence-level) orbital diagram, and write the condensed ground-state electron configuration for each: \begin{array}{ll}{\text { (a) } \mathrm{Mn}} & {\text { (b) } \mathrm{P}} & {\text { (c) Fe }}\end{array}
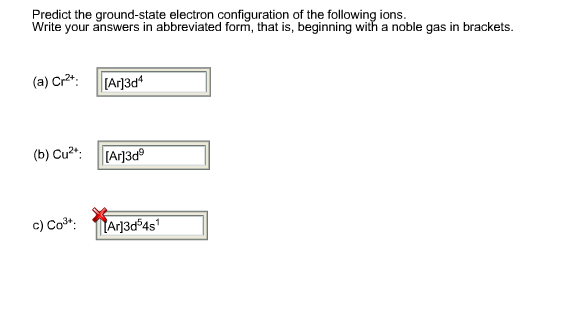
![SOLVED: Match each transition metal ion with its condensed ground-state electron configuration. Mn2+ [Ar]4s23d3 Hf2+ B [Kr]4d? Co3+ C [Ar]3d2 Fe3+ D [Xe]4fl45d2 Ag+ E Mo3+ [Kr]4dlo [Ar]3d6 G [Ar]3d5 SOLVED: Match each transition metal ion with its condensed ground-state electron configuration. Mn2+ [Ar]4s23d3 Hf2+ B [Kr]4d? Co3+ C [Ar]3d2 Fe3+ D [Xe]4fl45d2 Ag+ E Mo3+ [Kr]4dlo [Ar]3d6 G [Ar]3d5](https://cdn.numerade.com/ask_images/21363d649fc347378d9414a36d6f127c.jpg)